The One Big Beautiful Bill Act enacted in 2025 removed a prior policy exposure for drug developers focused on rare diseases. The legislation preserves a no-negotiation status for drugs that are approved for multiple rare indications which reduces a specific regulatory risk for smaller biotechnology firms represented in XBI. Prior policy allowed the loss of that protection when a drug expanded into additional uses which could have weakened expected revenue for affected developers.
The BIOSECURE Act reflects an administration emphasis on rebuilding the domestic drug supply chain. By penalizing reliance on foreign companies of concern the policy effectively favors US-based biotech infrastructure and alters the operating environment for domestic firms within XBI.
Restoration of Section 174 R&D expensing in 2026 permits eligible companies to deduct domestic research and development costs in the year incurred rather than amortizing those costs over several years. For pre-revenue companies in XBI this change lengthens available cash runway by a stated interval and reduces near-term pressure to access capital via dilutive offerings which can influence financing dynamics for the sector.
The MAHA movement has introduced regulatory headwinds that increase uncertainty for larger pharmaceutical firms and related market segments. Separately a push toward most favored nation pricing could compress margins for winners within the index which remains a material regulatory consideration for companies across the space.
This material is provided for informational and educational purposes only and does not constitute financial advice. All investments carry risk, including the potential loss of capital.
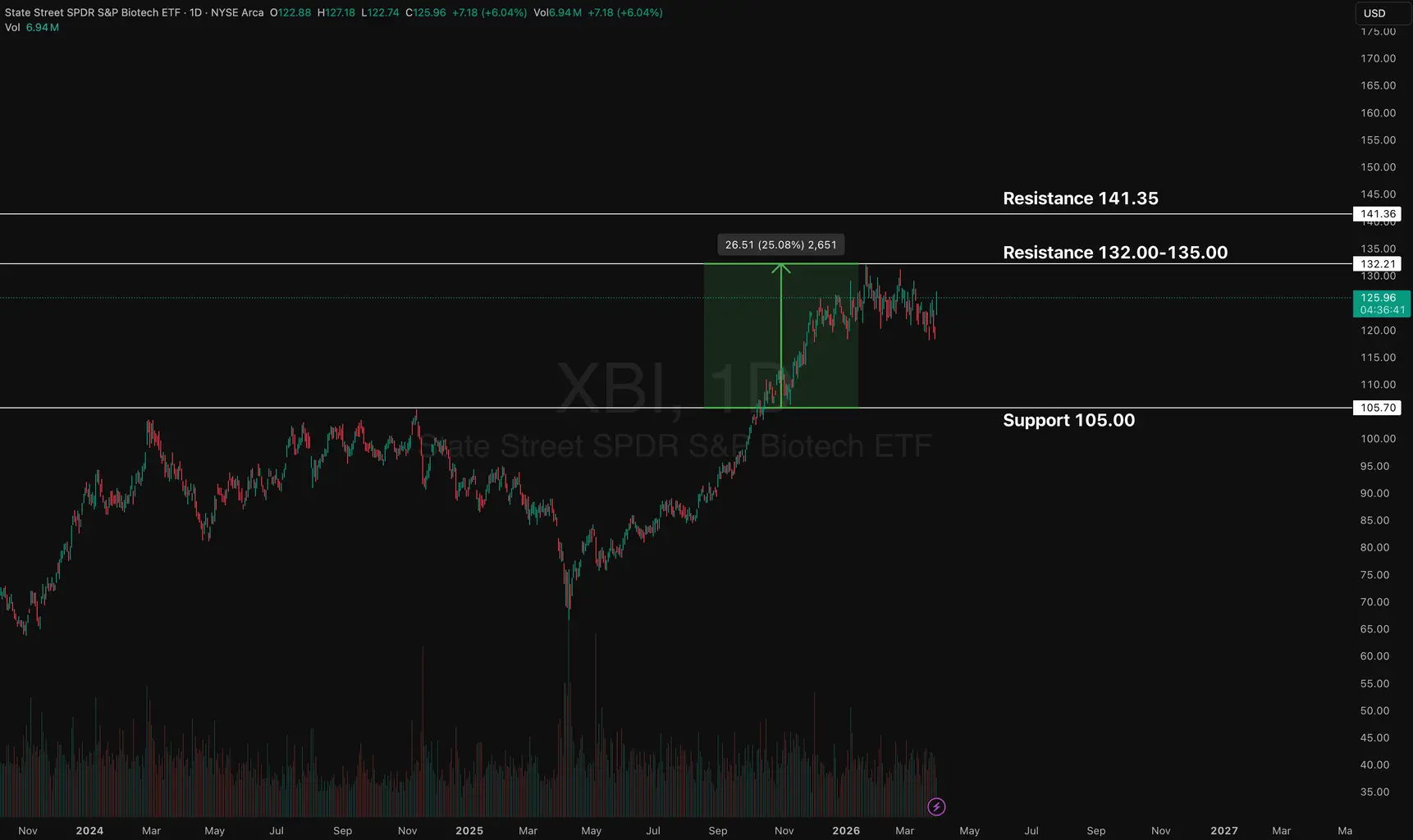
Feed